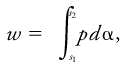Work: Difference between revisions
From Glossary of Meteorology
imported>Perlwikibot (Created page with " {{TermHeader}} {{TermSearch}} <div class="termentry"> <div class="term"> == work == </div> <div class="definition"><div class="short_definition">A form of energy a...") |
imported>Perlwikibot No edit summary |
||
| Line 9: | Line 9: | ||
</div> | </div> | ||
<div class="definition"><div class="short_definition">A form of [[energy]] arising from the motion of a system against a force, existing only in the process of [[energy conversion]].</div><br/> <div class="paragraph">Many forms of work (electrical, chemical, etc.) may be defined by analogy with mechanical work, but in meteorology the most frequently useful mathematical expression is that for the work per unit mass ''w'' done by a gaseous system in a given [[reversible process]] from thermodynamic state ''s''<sub>1</sub> to state ''s''<sub>2</sub>: <div class="display-formula"><blockquote>[[File:ams2001glos-We12.gif|link=|center|ams2001glos-We12]]</blockquote></div> where ''p'' is the [[pressure]] of the system and α its [[specific volume]]. The amount of work done will be a function of the particular process as well as of the initial and final states. <br/>''See'' [[heat]], [[first law of thermodynamics]].</div><br/> </div> | <div class="definition"><div class="short_definition">A form of [[energy]] arising from the motion of a system against a force, existing only in the process of [[energy conversion]].</div><br/> <div class="paragraph">Many forms of work (electrical, chemical, etc.) may be defined by analogy with mechanical work, but in meteorology the most frequently useful mathematical expression is that for the work per unit mass ''w'' done by a gaseous system in a given [[reversible process]] from thermodynamic state ''s''<sub>1</sub> to state ''s''<sub>2</sub>: <div class="display-formula"><blockquote>[[File:ams2001glos-We12.gif|link=|center|ams2001glos-We12]]</blockquote></div> where ''p'' is the [[pressure]] of the system and α its [[specific volume]]. The amount of work done will be a function of the particular process as well as of the initial and final states. <br/>''See'' [[heat]], [[first law of thermodynamics|first law of thermodynamics]].</div><br/> </div> | ||
</div> | </div> | ||
Latest revision as of 18:17, 25 April 2012
work
A form of energy arising from the motion of a system against a force, existing only in the process of energy conversion.
Many forms of work (electrical, chemical, etc.) may be defined by analogy with mechanical work, but in meteorology the most frequently useful mathematical expression is that for the work per unit mass w done by a gaseous system in a given reversible process from thermodynamic state s1 to state s2: where p is the pressure of the system and α its specific volume. The amount of work done will be a function of the particular process as well as of the initial and final states.
See heat, first law of thermodynamics.
See heat, first law of thermodynamics.