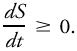Second law of thermodynamics
From Glossary of Meteorology
second law of thermodynamics
An inequality that is fundamentally different from the first law because it specifies the direction in which a natural process will evolve rather than merely requiring that certain quantities are conserved.
As formulated by Planck, the second law asserts that a thermodynamic state function, S, known as entropy, exists for all physical systems. For the universe and for a system isolated from its surroundings, Equality prevails only for reversible processes or when the system is in a steady state. When the universe of a system is a maximum, no further evolution of the system is possible. The second law is often asserted in other forms, including the following.
- When two bodies at different temperatures interact, the temperature of the hotter body can only decrease and that of the colder body can only increase unless work is done.
- No device can continuously deliver mechanical work and produce no effect other than cooling a reservoir.
- In the neighborhood of every state that can be reached reversibly, there exist states that cannot be reached by a reversible, adiabatic process, or, in other words, that can be reached only irreversibly or cannot be reached at all.
Dutton, J. A. 1995. Dynamics of Atmospheric Motion. Dover Press, . 45–51, 406–410.
Sommerfeld, A. 1964. Thermodynamics and Statistical Mechanics. Academic Press, . 26–36, 39.